 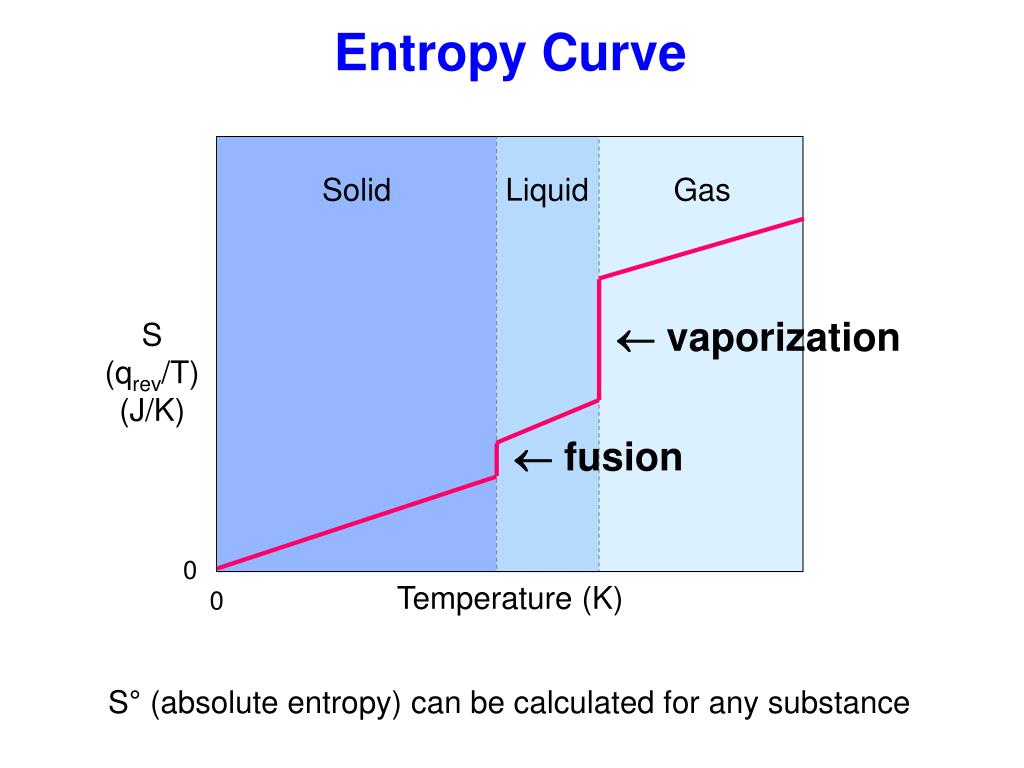 
For example, heat transfer cannot occur spontaneously. An important implication of this law is that heat transfers energy spontaneously from higher- to lower-temperature objects, but never spontaneously in the reverse direction. The total entropy of a system either increases or remains constant in any process it never decreases. The magnitude of the increase is greater than the magnitude of the decrease, so the overall entropy change for the formation of an NaCl solution is positive. The second law of thermodynamics states that the total entropy of a system either increases or remains constant in any spontaneous process it never decreases. The entropy change for a real, irreversible process is then equal to that for the theoretical reversible process that involves the same initial and final states. What is the decrease in entropy of 25.0 g of water that condenses on a bathroom mirror at a temperature of \(\displaystyle 35. Any chemical or physical change in a system may be accompanied by either an increase in entropy (S > 0) or a decrease in entropy (S. In 1865, Clausius named this property entropy (S) and defined its change for any process as the following: S qrev T (12.2.1) (12.2.1) S q rev T. Changes in entropy (S), together with changes in enthalpy (H), enable us to predict in which direction a chemical or physical change will occur spontaneously. Each hydrated ion, however, forms an ordered arrangement with water molecules, which decreases the entropy of the system. Entropy and the Second Law of Thermodynamics. Stated more generally, processes that locally decrease entropy, such as those that build and maintain the highly organized bodies of living things, can indeed take place. 
The system entropy will decrease when the system is dissolved in a solvent, such as when water is added to a solid. The system entropy will increase when the amount of motion within the system increases, such as when ice melts or evaporates. Differentiate between and determine the number of macrostates and microstates in a simple system of gas molecules, and explain why there is a higher probability that energy will be dispersed than it will be concentrated in a small number of nanoscale particles. To sum up, the high degree of organization of living things is maintained by a constant input of energy, and is offset by an increase in the entropy of the. Entropy is the amount of energy dispersal in a system. \) illustrates this procedure for the combustion of the liquid hydrocarbon isooctane (C 8H 18 2,2,4-trimethylpentane).\): The Effect of Solution Formation on Entropyĭissolving NaCl in water results in an increase in the entropy of the system. The net effect of the original process (local decrease in entropy) and the energy transfer (increase in entropy of surroundings) is an overall increase in the entropy of the universe.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
